Nanooze Blog
The world’s smallest QR code
Imagine this….credit cards have tiny super-secret chips inside them, just like the cool spy gadgets in movies. National mints print special marks on money to keep it safe, kind of like invisible ink. And places with super important stuff, like top-secret labs, use scanners that look at your eyes to make sure you’re supposed to be there.

Brochosomes are 3D printed structures that can carry information
Now, there are some sneaky people out there who try to steal this stuff. But don’t worry! Sheng Shen and his colleagues at Penn State University have come up with a super clever idea. They’re making pictures that hide secrets pixel by pixel, like magic! They want to use this trick to make things even safer from sneaky thieves and make sure nobody can copy important stuff. Cool, huh?
They use these things called brochosomes. Brochosomes are like tiny soccer balls that have super tiny holes inside them. They don’t reflect light like normal things; instead, they soak it up! Scientists think this helps leafhoppers hide in the world around them, like sneaky little ninjas!
They make brochosomes with a3D printer that can make tiny things! So, there’s this team from Penn State who figured out how to make a special kind of code called a QR code. But get this, it’s so tiny, you can only see it with a microscope! They made it using a fancy printer that can control if each little dot in the code has holes or not. And guess what? You can only read this code with a special camera that sees in infrared light! Isn’t that neat? They’re thinking about making bigger ones for everyday stuff too. How cool is that?
Blowin’ in the wind.
The title is a Bob Dylan song. But in this case we are talking about renewal energy and generating electricity by harnessing the wind. Nanotechnology c

Wind turbine
an be used to create new materials with amazing functions. Wind turbines are a fairly common sight these days, enormously wind mills with blades that turn slowly in the wind. These blades need to be light and strong. Light because the lighter the blade the less wind power it takes to move it. Strong because it needs to work a long time and a wind turbine might face strong winds. Cost is also a factor how much the materials cost and how durable the wind turbines.
Enter nanotechnology. Not only can nanotechnology be used to create the next-generation of wind turbines but also help to produce coatings that reduce drag and make the blades move through the air more efficiently. One area of focus is in direct drive wind turbines which eliminate the need for a gear box. Gear boxes are needed to match the rotation speed of the blades with the electrical generator. Nanotechnology will also impact the development of new sensors to monitor the energy being generated by the wind turbine.
Read more here
New nano-thing to detect disease
Disease detection is a never ending challenge and the goal is to create new tests for diseases that are faster, cheaper and easier. Tests that are slow, expensive and tough to do, don’t get done as often and diseases that might be cured are not cured. Nanotechnology to

The inventor of QolorEX platform, Professor Sara Mahshid
the rescue! Scientists at McGill University have invented a new approach called the QolorEX platform which can be done with saliva samples (no blood letting necessary) and the results analyzed with a smart phone. The QolorEX platform uses DNA amplification and a lot of fancy nanometer-sized fluid handling components to process the sample and image the results. The goal is to make a disease detection system that can be used in low-resource areas where health care is a problem but there is a lot of need.
Zapping those summer pests
It is Summer and time to be outside. But there are those pesky mosquitoes that are attracted to us and bite sometimes also passing along viral diseases. Nanotechnology to the rescue. A team of researchers have invented a new self-powered generator that can zap mosquitoes. These nanometer-sized “tribioelectric nanogenerators (TENGs) can produce t

heir own energy and use that energy to zap mosquitoes sort of like those bug zappers that are so entertaining (until you get a giant moth and the whole thing almost catches on fire). The scientists used a combination of new theoretical model and some dandy ferroelectric nanocomposites to create a TENG that generated about 6 kV and that could power close to 3500 LED. The new bug zapper is a ways away from being a commercial product but stay tuned.
Over the lips and past the gums….
Problems with your gastrointestinal tract are no laughing matter. Some are serious and can cause a life-long struggle that impacts the foods we can eat. And so no fun. Taking medications is not new but new approaches to solving the problems with your gastrointestinal tract are on the way. Nanotechnology offers the promise of better ways of diagnosing and treating gastrointestinal diseases. Part of the problem is getting past the sto mach (and being able to simply swallow something. Researchers at the University of Maryland have created a digestible capsule that can pass through the stomach and then be able to carry out tasks in the gastrointestinal tract. One target is inflammatory bowel disease which affects 3 million Americans. The new material can be fabricated into different shapes and sizes, perhaps the next space ship for a Fantastic Voyage. For more information look here
mach (and being able to simply swallow something. Researchers at the University of Maryland have created a digestible capsule that can pass through the stomach and then be able to carry out tasks in the gastrointestinal tract. One target is inflammatory bowel disease which affects 3 million Americans. The new material can be fabricated into different shapes and sizes, perhaps the next space ship for a Fantastic Voyage. For more information look here
Vaccines that don’t need ultracold
The new crop of vaccines for COVID that use something called mRNA usually require super cold storage. That makes it hard to distribute these vaccines especially in places that don’t have a lot of ultra-low-temperature refrigeration. The mRNA vaccine particles are coated in lipids and less than 100 nanometers in size. Scientists in a publication called SMALL (yes, this is no lie, the journal is called SMALL) reported testing three different drying processes to help preserve these vaccines. The experiments show that these mRNA vaccines could be preserved through a process much like freeze drying but also included a sugar, trehalose which helped to keep these vaccines stable at room temperature. While nobody likes a shot, they are sometimes important and making sure that they can be shipped and used all around the world is very important.
Edible nanoparticles-yummmm
Most food is perishable, and food that spoils before it is consumed is a waste. Plus foods can sometimes contain dangerous microbes that can make us sick. Enter nanotechnology. There are a lot of compounds that can inhibit the growth of microorganisms in foods, but keeping them active is a challenge. Scientists from China have been using nanotechnology to make little capsules that can carry compounds like nisin (a natural antibiotic) and rosemary oil. Particles are about 200 nanometers in size and could reduce microbial numbers by 100 times or more without a yucky taste.
Too much information
 We don’t often think about the toilet as a source of information, but scientists at Cambridge University are developing an ‘intelligent loo’ that is able to analyze your urine. The toilet is engineered to contain optical sensors that detect different chemicals in your urine. The data is then uploaded to your phone (but hopefully not your Facebook page) indicating potential problems or perhaps instructions to adjust the dosage of a medication. The field of personalized medicine continues to explode and an one-size fits all approach to medicine is being replaced by treatments tailored for each individual.
We don’t often think about the toilet as a source of information, but scientists at Cambridge University are developing an ‘intelligent loo’ that is able to analyze your urine. The toilet is engineered to contain optical sensors that detect different chemicals in your urine. The data is then uploaded to your phone (but hopefully not your Facebook page) indicating potential problems or perhaps instructions to adjust the dosage of a medication. The field of personalized medicine continues to explode and an one-size fits all approach to medicine is being replaced by treatments tailored for each individual.
Take two and call me in the morning.
Nanotechnology offers new ways of diagnosing diseases. Most tests require taking a sample, like blood, or urine and then sending them

to a laboratory. Scientists at the Massachusetts Institute of Technology ha
ve developed a sensor that you can swallow. Inside of the sensor are bacteria which have been engineered to glow if they are exposed to a certain chemical. The glowing bacteria then send out a radio signal which is detected using a smart phone. The pill was engineered to detect blood in the gastrointestinal tract, a sign of ulcers. The pill is part biology and part engineering. What happens to the pill after you ingest it is obvious and no information was provided about if this is pill is recycled or well down the drain it goes.
Sometimes we do nano for fun
Researchers at Georgia Tech have made the ‘World’s Smallest Ad’ created for Arby’s, the fast food restaurants.  The ad was printed on the side of a sesame seed, like the one you might find….wait for it….on a roast beef sandwich bun. The size of the ad was about 800 square microns (less than the width of a hair in either dimension) which is pretty small but still almost a billion square nanometers. If you were at the Arby’s in NYC apparently you got to ‘see’ one of these seeds using a scanning electron microscope. What happened to this seed isn’t obvious, hopefully it didn’t wind up on a roast beef sandwich that was woofed down by a customer.
The ad was printed on the side of a sesame seed, like the one you might find….wait for it….on a roast beef sandwich bun. The size of the ad was about 800 square microns (less than the width of a hair in either dimension) which is pretty small but still almost a billion square nanometers. If you were at the Arby’s in NYC apparently you got to ‘see’ one of these seeds using a scanning electron microscope. What happened to this seed isn’t obvious, hopefully it didn’t wind up on a roast beef sandwich that was woofed down by a customer.
Medicine release on demand
Some medicines are more effective when they are delivered at the site where they are needed and when they are needed. Think about taking an aspirin but it works only when you have a headache. Scientists from 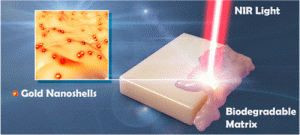 have developed a biodegradable material with nanometer-scale gold particles that help release medicines embedded in the material. When you shine an infrared light on the material, the gold particles heat up the biodegradable material and release the medicine. Near infrared-wavelength light is used because it can penetrate a lot deeper into the body than regular visible light. These biodegradable materials might be implanted into the body and then release important medicines on demand
have developed a biodegradable material with nanometer-scale gold particles that help release medicines embedded in the material. When you shine an infrared light on the material, the gold particles heat up the biodegradable material and release the medicine. Near infrared-wavelength light is used because it can penetrate a lot deeper into the body than regular visible light. These biodegradable materials might be implanted into the body and then release important medicines on demand
Sometimes is it just a great picture
Nanotechnology has contributed to the advances in our ability to see different things at the nanoscale. Microscopy has advanced from the very early days of microscopes being a single glass lens to very advanced instruments with nanometer resolution. We can see lots of stuff with high resolution and even in 3D. And sometimes the picture is just neat. Scientists at St. Judes Hospital used a confocal microscope to study the progression of a soft tissue cancer to understand the origins of these cancer cells. It was once believed these cancer cells came from muscle tissue but in fact are from cells that make up blood vessels. The technique and the image involve immunostaining, using antibodies against bind to different things. The antibodies are ‘painted’ with a dye that results in different colors. The cool image may help doctors understand
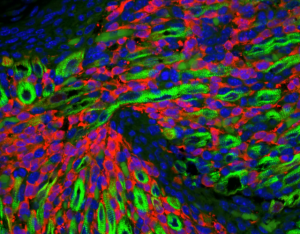 how an important disease like cancer develops and how it might be cured.
how an important disease like cancer develops and how it might be cured.
You could mow your lawn at night
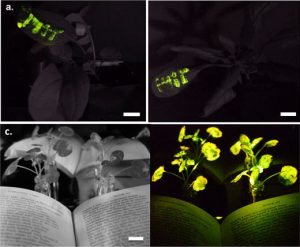
Scientists from MIT and the University of California have figured out a way to engineer plants to glow in the dark. This isn’t the first time but it is the first time that it has been on whole plants without initially do some tricky genetics. They use nanoparticles that introduce the same components that fireflies use to glow in the dark. These tiny particles are forced into plant cells by pressure and once there they make the plants glow. There are lots of applications but we kind of like the idea of being able to mow in the dark. OK, that is a joke.
May the force be with you
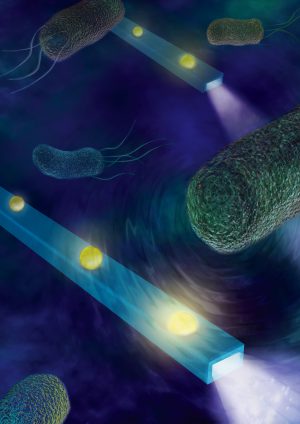
Mostly everything has a nano-unit measurement including sounds. The human ear can hear things down to around 0 decibels. If you are about 100 feet from a jet as it takes off that is about 150 decibels. Your headphones can be cranked up to around 100 decibels. Scientists at the University of California in San Diego have made nano-fibers that are sensitive enough to hear things at -30 decibels. That is around 1000 times more sensitive than the human ear (decibels are measured on a log scale). They can use this device to ‘hear’ heart muscles beat. These scientists imagine that their device might be used to ‘hear’ a single bacteria (sneaking up behind you) or changes in cells that might signal their becoming cancerous. Their work was supported by the National Science Foundation
Flexing your mussel
Before you call the typo police, we are talking mussels, not muscles. Researchers at Purdue University have developed an adhesive that is based upon the same stuff that mussels use to stay stuck to wooden poles, rocks and other places that mussels like to hang out. They claim the synthetic mussel-based glue is 10 times stronger than commercial glues. These researchers studied the components of the goo that helps mussels stick to things and then made their own glue based upon the molecules in this natural material. The secret sauce is a molecule called DOPA which is an amino acid (one of the building blocks of proteins) and that is what makes the mussel glue so strong. The mussel-based glue even works underwater (well of course).
For a video click here 
Float like a drone, pollinate like a bee
Bees carry out important work by pollinating flowers—they move pollen from one part of the flower to another or between flowers. They contribute something like $29 billion dollars to the farm economy in the US alone. For a number of reasons the bee population is in decline and that is bad. One solution to the loss of these important pollinators are tiny drones that are built to go from flower to flower where they pick up and drop off pollen. Scientists in Japan lead by Eijiro Miyako at the National Institute of Advanced Industrial Science and Technology (AIST) Nanomaterial Research Institute have designed and tested a tiny drone that can pollinate flowers. The drone has material that acts like the tiny hairs on a bee and collect the pollen then deposit it elsewhere. We hope that there is a way to save the real bees but in the meantime this is a cool solution.
Nanotechnology and noodles
Sometimes science can just be fun if not edible. Scientists at MIT have developed a process to make pasta that shape-shifts upon cooking. They claim it could save on shipping costs because you might be able to pack these flat noodles into a smaller container (think lasagna noodles stacked up, vs elbows). Fair enough. The real fun is that by adding layers of materials, the pasta can be made to curl up and form different shapes upon being dunked into hot water. They use layers of different materials like gelatin and cellulose and 3D print them. More important is their ability to predict the shape based upon the process or more to make a shape by design and then fabricate it.
Even smaller!

Max Shulaker a grad student at Stanford University shows off the new material used to make new computers on Monday August 19, 2013 at the Stanford School of Engineering. ( Photo by Norbert von der Groeben )
Making computer parts smaller and smaller is the reason why your average laptop is a zillion times more powerful than computers from 50 years ago that used to fill up an entire room. The basic component of a computer chip is a transistor which is a switch that turns off and on. Today computer chips have close to two billion (2,000,000,000) transistors and counting. New materials including carbon nanotubes are being used to build different kinds of transistors. The first carbon nanotube transistors were made a few years ago (2013) at Stanford University and story was recently made into a video. This work was supported by the National Science Foundation
See the video here
Sometimes you just do it for fun
The world’s smallest version of the Edmonton Oilers logo has been created by a group of scientists at the University of Alberta. The Oilers are the city’s NHL hockey team and they are currently in the Stanley Cup playoffs. The logo is only 2400 nanometers which is about twice the size of your average bacteria. About 900 million of these logos would fit on a hockey puck (think about that for a bit). The last time they made a nano-logo was 2006, unfortunately the Oilers lost to the Carolina Hurricanes, so this logo, about 40 times smaller is hopefully going to lead to a better outcome.
Shining light on nanocubes
Methane is the building block of a lot of different fuels. There are a variety of methanes sources (think cows!) but on source of methane is to make it from carbon dioxide. There is lots of carbon dioxide but converting it to methane requires energy. Scientists at Duke University have developed a process that uses ultraviolet light and nanocubes made out of rhodium. Rhodium is an element that is pretty rare and the naocubes are about 37 nanometers on a side. When the UV light shines on the rhodium nanocubes the energy from the light helps to convert carbon dioxide into methane. The reaction is pretty specific and there aren’t a lot of other products besides methane. One byproduct they are trying to avoid is carbon monoxide. Taking carbon dioxide out of the atmosphere while making fuel is a neat way to help reduce global warming and also provide a renewable energy source.
This work was supported by the National Science Foundation along with Army Research Office and the Department of Energy.